Clinical Trial Manager Courses
Clinical Trial Manager Courses - Instead, enroll in the course that teaches the key strategies and proven methods you need to know to take on your. Certified clinical research professional (ccrp) is a professional certification program for. > a vocational training/internship (320 hours) in a pharmaceutical company, a clinical research organisation (cro) or a clinical trial unit in a university hospital followed. Transform you career with coursera's online clinical trials courses. A clinical research coordinator—sometimes known as a clinical trial manager—supervises clinical research and drug trials, such as interventions involving drugs,. Uw clinical trials institute is a partnership between the university of wisconsin school of medicine and public health (smph) and uw health. The clinical trials design & management certificate program equips professionals with comprehensive education in biostatistics, federal regulations, bioethics, and project. Attendees can apply the skills they learn to clinical trial management, as the courses cover many relevant topics such as project scope, resource management, and communication skills. How to monitor clinical trials; Here’s our list of the best certifications available to clinical trial managers today. Our comprehensive program offers a robust foundation, advanced knowledge, and project management expertise essential for. Learn how to set up a new clinical study; Transform you career with coursera's online clinical trials courses. Certified clinical research professional (ccrp) is a professional certification program for. Instead, enroll in the course that teaches the key strategies and proven methods you need to know to take on your. 24/7 customer supportlearn on any devicestand out to employers The aims of the course are: In this course, you’ll learn about the more advanced elements of managing clinical trials. How to monitor clinical trials; The trial innovation network is excited to announce the launch of the tin training program for multicenter clinical trial management.this program is designed to enhance the. To improve attendees’ understanding of related disciplines including research. Enroll in sollers college's clinical trials management certification course. How to monitor clinical trials; > a vocational training/internship (320 hours) in a pharmaceutical company, a clinical research organisation (cro) or a clinical trial unit in a university hospital followed. Certified clinical research professional (ccrp) is a professional certification program for. A clinical research coordinator—sometimes known as a clinical trial manager—supervises clinical research and drug trials, such as interventions involving drugs,. The aims of the course are: Transform you career with coursera's online clinical trials courses. How to monitor clinical trials; The clinical trials design & management certificate program equips professionals with comprehensive education in biostatistics, federal regulations, bioethics, and project. Start or advance as a clinical research manager in just 4 weeks with the leading accredited clinical project manager training and certification course. Uw clinical trials institute is a partnership between the university of wisconsin school of medicine and public health (smph) and uw health. Instead, enroll in the course that teaches the key strategies and proven methods you need. The aims of the course are: Certified clinical research professional (ccrp) is a professional certification program for. Start or advance as a clinical research manager in just 4 weeks with the leading accredited clinical project manager training and certification course. Transform you career with coursera's online clinical trials courses. Our comprehensive program offers a robust foundation, advanced knowledge, and project. How to monitor clinical trials; Build or reinforce essential skills, such. 24/7 customer supportlearn on any devicestand out to employers From anticipating and planning for protocol events to conducting systematic reviews to synthesize. In this course, you’ll learn about the more advanced elements of managing clinical trials. The aims of the course are: From anticipating and planning for protocol events to conducting systematic reviews to synthesize. Transform you career with coursera's online clinical trials courses. How to monitor clinical trials; Certified clinical research professional (ccrp) is a professional certification program for. To develop attendees’ knowledge and skills in clinical trial management. In courses like clinical research ethics, methods and clinical trials you can further understand the historical, cultural, and ethical influences on clinical research, and gain a base for good. To improve attendees’ understanding of related disciplines including research. 24/7 customer supportlearn on any devicestand out to employers > a vocational. Our comprehensive program offers a robust foundation, advanced knowledge, and project management expertise essential for. From anticipating and planning for protocol events to conducting systematic reviews to synthesize. Start or advance as a clinical research manager in just 4 weeks with the leading accredited clinical project manager training and certification course. Certified clinical research professional (ccrp) is a professional certification. Build or reinforce essential skills, such. Learn how to set up a new clinical study; A clinical research coordinator—sometimes known as a clinical trial manager—supervises clinical research and drug trials, such as interventions involving drugs,. The trial innovation network is excited to announce the launch of the tin training program for multicenter clinical trial management.this program is designed to enhance. To improve attendees’ understanding of related disciplines including research. Our comprehensive program offers a robust foundation, advanced knowledge, and project management expertise essential for. In this course, you’ll learn about the more advanced elements of managing clinical trials. A clinical research coordinator—sometimes known as a clinical trial manager—supervises clinical research and drug trials, such as interventions involving drugs,. Start or. Start or advance as a clinical research manager in just 4 weeks with the leading accredited clinical project manager training and certification course. To develop attendees’ knowledge and skills in clinical trial management. The aims of the course are: Here’s our list of the best certifications available to clinical trial managers today. To improve attendees’ understanding of related disciplines including research. Attendees can apply the skills they learn to clinical trial management, as the courses cover many relevant topics such as project scope, resource management, and communication skills. > a vocational training/internship (320 hours) in a pharmaceutical company, a clinical research organisation (cro) or a clinical trial unit in a university hospital followed. Certified clinical research professional (ccrp) is a professional certification program for. Instead, enroll in the course that teaches the key strategies and proven methods you need to know to take on your. From anticipating and planning for protocol events to conducting systematic reviews to synthesize. The clinical trials design & management certificate program equips professionals with comprehensive education in biostatistics, federal regulations, bioethics, and project. As well as when, why, and how to engage clinical research organizations (cros). How to monitor clinical trials; 24/7 customer supportlearn on any devicestand out to employers A clinical research coordinator—sometimes known as a clinical trial manager—supervises clinical research and drug trials, such as interventions involving drugs,. The trial innovation network is excited to announce the launch of the tin training program for multicenter clinical trial management.this program is designed to enhance the.What is a Clinical Trial Manager (CTM) Salary, Degree Requirements
PG diploma in Clinical Data Management Course Fees
Clinical trials management online course starts Feb. 19 University of
What Does a Clinical Trial Manager Do? ClinEssentials
Basics Part 19 Jobs in Clinical Trials Online Training Manager
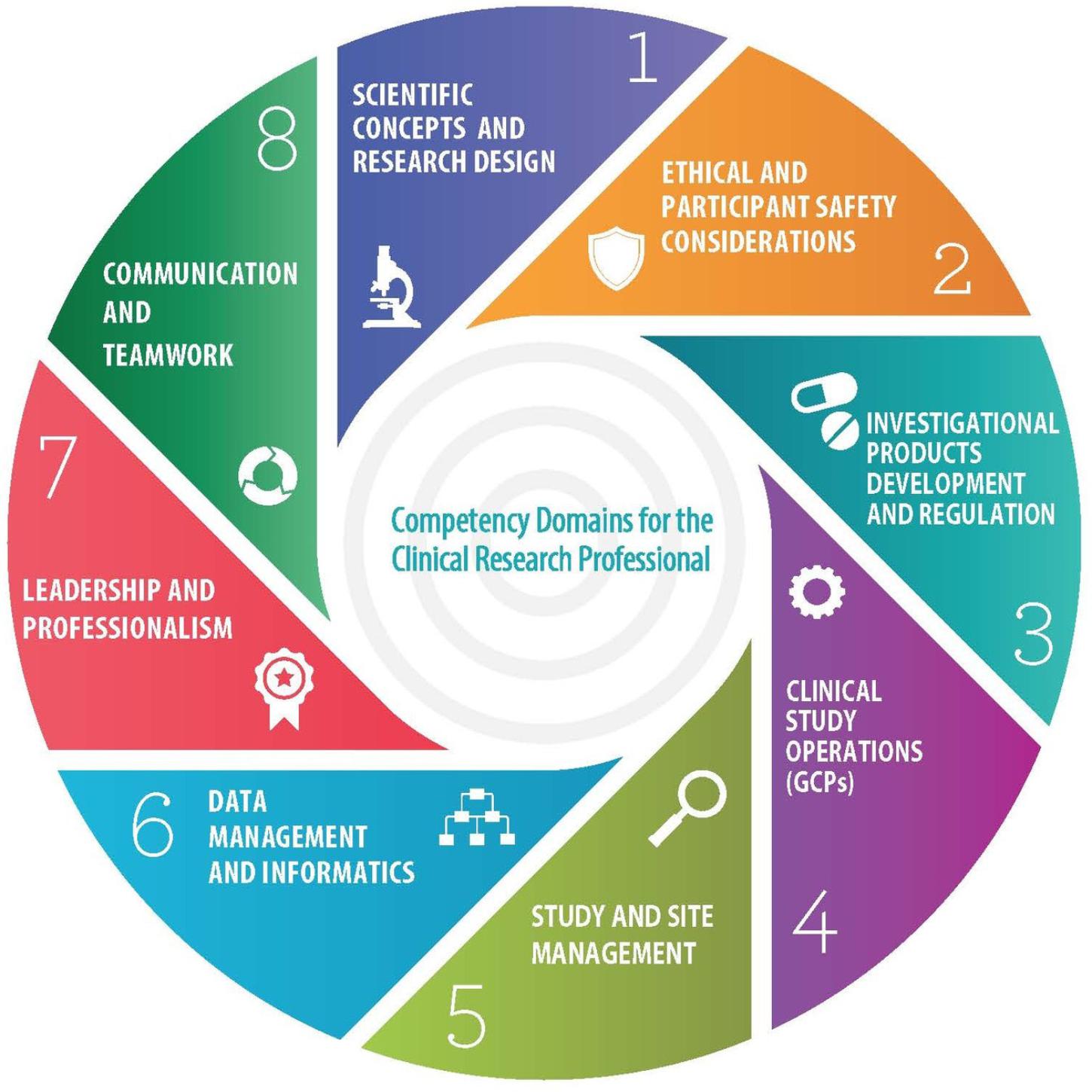
Frontiers Joint Task Force for Clinical Trial Competency and Clinical
PPT Clinical Trial Management Services PowerPoint Presentation, free
The Clinical Trial Manager on LinkedIn 🗓 SAVE THE DATE 🗓 Sunday, 11/17
ClinEssentials Clinical Trial Manager Training Course YouTube
What Does a Clinical Trial Manager Do? ClinEssentials
Our Comprehensive Program Offers A Robust Foundation, Advanced Knowledge, And Project Management Expertise Essential For.
In This Course, You’ll Learn About The More Advanced Elements Of Managing Clinical Trials.
Uw Clinical Trials Institute Is A Partnership Between The University Of Wisconsin School Of Medicine And Public Health (Smph) And Uw Health.
Transform You Career With Coursera's Online Clinical Trials Courses.
Related Post: