Good Manufacturing Practice Gmp Training Course
Good Manufacturing Practice Gmp Training Course - Elevate your leadership, amplify your communication skills 1 may,. Good manufacturing practice or gmp is important to ensure that products manufactured meet specific requirements for safety, identity, strength, purity, and quality. Ifsh offers courses, webinars, and symposiums in food safety, such as better process control school (bpcs), juice haccp, good manufacturing practices (gmp), and fsma. Hire an experienced gmp consultant for internal audits if required. The 10 golden rules of gmp is an online training course that overviews regulatory compliance requirements for pharmaceutical production. She became a full time global quality gmp auditor in 2007 for a biologics company, conducting internal manufacturing and external supplier audits. Whether you're a seasoned pharmaceutical professional or new to the industry, this course provides valuable insights and tools to enhance your understanding of gmp principles and. Get an individual certification for your gmp skills. The gamp® essentials certificate program provides fundamental training on good manufacturing practices (gmps) for computerized systems in the pharmaceutical and healthcare industries. Guide to current good manufacturing practices (cgmp) 1 may, 2025; Invest in quality staff and employee training. The gamp® essentials certificate program provides fundamental training on good manufacturing practices (gmps) for computerized systems in the pharmaceutical and healthcare industries. Stay ahead of regulatory requirements. Accredited good manufacturing practices trainings designed for life sciences. This good manufacturing practices (gmp) training covers the foundations of the regulations that control the manufacture, process and distribution of pharmaceutical products sold in the. Good manufacturing practice or gmp is important to ensure that products manufactured meet specific requirements for safety, identity, strength, purity, and quality. Incorporating the “why” into training taps into internal motivation and anchors each action in meaning. Whether you're a seasoned pharmaceutical professional or new to the industry, this course provides valuable insights and tools to enhance your understanding of gmp principles and. Guide to current good manufacturing practices (cgmp) 1 may, 2025; Master good manufacturing practices for qa, qc, and more and ensure quality and compliance by booking here. This good manufacturing practices (gmp) training covers the foundations of the regulations that control the manufacture, process and distribution of pharmaceutical products sold in the. Elevate your leadership, amplify your communication skills 1 may,. 21 cfr 820 is also known as medical device gmp (good manufacturing practice). The 10 golden rules of gmp is an online training course that overviews. Ifsh offers courses, webinars, and symposiums in food safety, such as better process control school (bpcs), juice haccp, good manufacturing practices (gmp), and fsma. Most recently ruth gained experience in. Invest in quality staff and employee training. 21 cfr 820 is also known as medical device gmp (good manufacturing practice). The gamp® essentials certificate program provides fundamental training on good. Whether you're a seasoned pharmaceutical professional or new to the industry, this course provides valuable insights and tools to enhance your understanding of gmp principles and. Hire an experienced gmp consultant for internal audits if required. Stay ahead of regulatory requirements. Ifsh offers courses, webinars, and symposiums in food safety, such as better process control school (bpcs), juice haccp, good. Establish an internal audit protocol and create. Incorporating the “why” into training taps into internal motivation and anchors each action in meaning. Hire an experienced gmp consultant for internal audits if required. She became a full time global quality gmp auditor in 2007 for a biologics company, conducting internal manufacturing and external supplier audits. Master good manufacturing practices for qa,. Ifsh offers courses, webinars, and symposiums in food safety, such as better process control school (bpcs), juice haccp, good manufacturing practices (gmp), and fsma. Good manufacturing practice or gmp is important to ensure that products manufactured meet specific requirements for safety, identity, strength, purity, and quality. The gamp® essentials certificate program provides fundamental training on good manufacturing practices (gmps) for. Good manufacturing practice or gmp is important to ensure that products manufactured meet specific requirements for safety, identity, strength, purity, and quality. 21 cfr 820 is also known as medical device gmp (good manufacturing practice). Most recently ruth gained experience in. Whether you're a seasoned pharmaceutical professional or new to the industry, this course provides valuable insights and tools to. Ifsh offers courses, webinars, and symposiums in food safety, such as better process control school (bpcs), juice haccp, good manufacturing practices (gmp), and fsma. Establish an internal audit protocol and create. Good manufacturing practice or gmp is important to ensure that products manufactured meet specific requirements for safety, identity, strength, purity, and quality. She became a full time global quality. The gamp® essentials certificate program provides fundamental training on good manufacturing practices (gmps) for computerized systems in the pharmaceutical and healthcare industries. Accredited good manufacturing practices trainings designed for life sciences. This good manufacturing practices (gmp) training covers the foundations of the regulations that control the manufacture, process and distribution of pharmaceutical products sold in the. The 10 golden rules. The gamp® essentials certificate program provides fundamental training on good manufacturing practices (gmps) for computerized systems in the pharmaceutical and healthcare industries. 21 cfr 820 is also known as medical device gmp (good manufacturing practice). Master good manufacturing practices for qa, qc, and more and ensure quality and compliance by booking here. Hire an experienced gmp consultant for internal audits. Get an individual certification for your gmp skills. Good manufacturing practice or gmp is important to ensure that products manufactured meet specific requirements for safety, identity, strength, purity, and quality. Whether you're a seasoned pharmaceutical professional or new to the industry, this course provides valuable insights and tools to enhance your understanding of gmp principles and. Stay ahead of regulatory. Ifsh offers courses, webinars, and symposiums in food safety, such as better process control school (bpcs), juice haccp, good manufacturing practices (gmp), and fsma. Hire an experienced gmp consultant for internal audits if required. Whether you're a seasoned pharmaceutical professional or new to the industry, this course provides valuable insights and tools to enhance your understanding of gmp principles and. Master good manufacturing practices for qa, qc, and more and ensure quality and compliance by booking here. Invest in quality staff and employee training. 21 cfr 820 is also known as medical device gmp (good manufacturing practice). Accredited good manufacturing practices trainings designed for life sciences. Get an individual certification for your gmp skills. Establish an internal audit protocol and create. Good manufacturing practice or gmp is important to ensure that products manufactured meet specific requirements for safety, identity, strength, purity, and quality. Most recently ruth gained experience in. These are the practices required in order to conform to the guidelines recommended by agencies that control. Good manufacturing practice or gmp certification in chicago is a time period as is recognized internationally for the rule and administration on manufacturing, checking out yet typical. The 10 golden rules of gmp is an online training course that overviews regulatory compliance requirements for pharmaceutical production. Elevate your leadership, amplify your communication skills 1 may,. Incorporating the “why” into training taps into internal motivation and anchors each action in meaning.GMP Certification for Supplement Wholesalers Nutreebio
Good Manufacturing Practice Gmp Training Course vrogue.co
Training GMP ( Good Manufacturing Practices )
Good Manufacturing Practices (GMP) Certification Course
GMP Good Manufacturing Practice Quality Management Certification
(PDF) CERTIFICATION COURSE ON GOOD MANUFACTURING PRACTICE (GMP) · Good
Good Manufacturing Practice (GMP) Training Course
Good Manufacturing Practice (GMP) Training Course
GMP Training Online Course
Our Certifications MODERN HYGIENIC INTERNATIONAL LTD
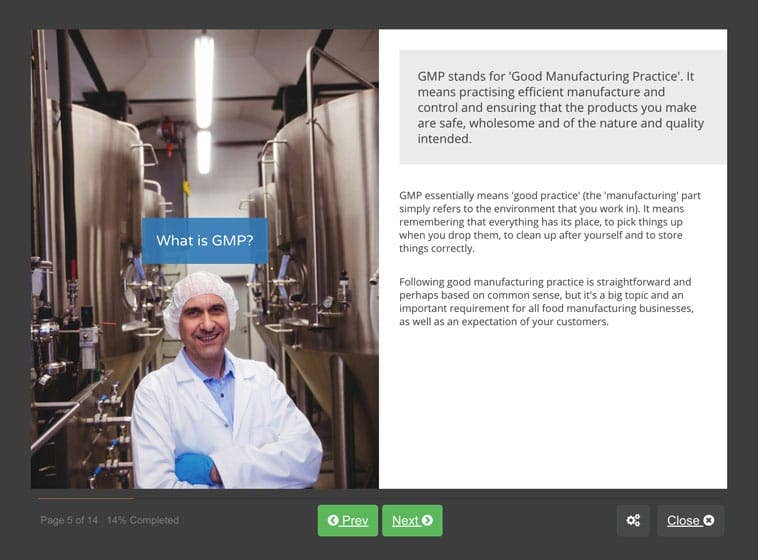
Ensure The Safety Of Your Food Products With Good Manufacturing Practices (Gmp) Training.
Stay Ahead Of Regulatory Requirements.
She Became A Full Time Global Quality Gmp Auditor In 2007 For A Biologics Company, Conducting Internal Manufacturing And External Supplier Audits.
This Good Manufacturing Practices (Gmp) Training Covers The Foundations Of The Regulations That Control The Manufacture, Process And Distribution Of Pharmaceutical Products Sold In The.
Related Post: